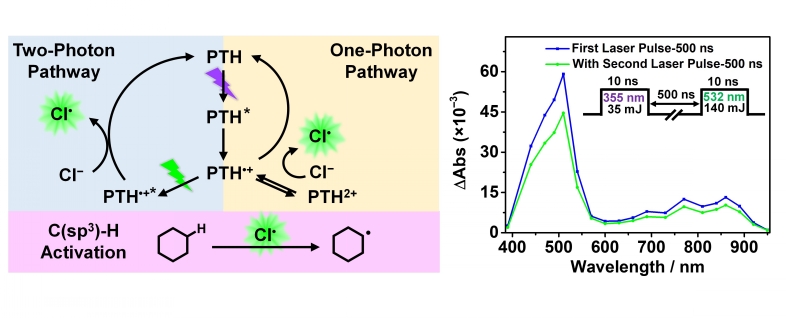
Chloride oxidation has tremendous utility in the burgeoning field of chlorine-mediated C–H activation, yet it remains a challenging process to initiate with light because of the exceedingly positive one-electron reduction potential, E° (Cl•/–), beyond most common transition-metal photooxidants. Herein, two photocatalytic chloride oxidation pathways that involve either one- or consecutive two-photon excitation of N-phenylphenothiazine (PTH) are presented. The one-photon pathway generates PTH•+ by oxidative quenching that subsequently disproportionates to yield PTH2+ that oxidizes chloride; this pathway is also accessed by the electrochemical oxidation of PTH. The two-photon pathway, which proceeded through the radical cation excited state, 2PTH•+*, was of particular interest as this super-photooxidant was capable of directly oxidizing chloride to chlorine atoms. Laser flash photolysis revealed that the photooxidation by the doublet excited state proceeded on a subnanosecond timescale through a static quenching mechanism with an ion-pairing equilibrium constant of 0.36 M–1. The PTH photoredox chemistry was quantified spectroscopically on nanosecond and longer time scales, and chloride oxidation chemistry was revealed by reactivity studies with model organic substrates. One- and two-photon excitation of PTH enabled chlorination of unactivated C(sp3)–H bonds of organic compounds such as cyclohexane with substantial yield enhancement observed from inclusion of the second excitation wavelength. This study provides new mechanistic insights into chloride oxidation catalyzed by an inexpensive and commercially available organic photooxidant.
https://pubs.acs.org/doi/10.1021/jacs.2c07107